|
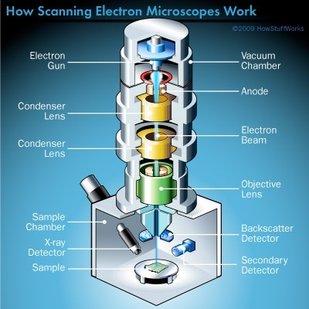
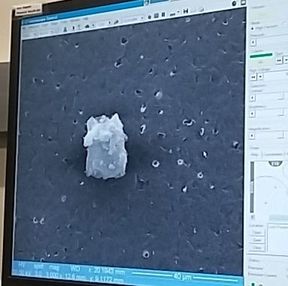
Remember back in week 2 when I mentioned that there is a possibility for the biodegradable polymer to activate platelets? That was what my week circled around. We needed to find out how many platelets were activated due to the presence of the polymer, so after a static test in which plasma (with platelets and the proteins that allow them to aggregate) was allowed to rest on the polymer, we made our way to a different lab (field trip!). This new lab was impressive, with many machines larger than I am (granted, not a tough height to beat). The downside? I was probably not allowed to be there, so I maintained a low profile and told myself to touch nothing. In order to see how many platelets had activated and aggregated, we used a scanning electron microscope (SEM). This microscope sends a beam of electrons onto the surface of the object, and detectors create a signal off of the back-scattered electrons, x-rays, and secondary electrons that are ejected from the sample after the beam hits the sample's atoms. The computer then uses this signal to create a detailed image of the topography of the sample. Before we could get to mapping out the surface of the polymer, we had to prepare our samples. First, we cut up our polymer samples and placed them on little discs compatible with the SEM. (This part was rougher than I had anticipated - apparently I am not as nimble with double sided tape as I had originally thought. Needless to say, this internship has been an incredibly humbling experience.) Our next step was probably the most exciting. Here is where goldsmithing met chemical science. When an electron beam is focused on non-conductive material (such as platelets or polymers), there is a possibility for the subject to become charged. This can cause the microscope to misread the surface and create an inaccurate image. The solution? Sputter-coat the surface of the polymer in gold. To allow this process to take place, a few steps need to occur. First, you increase the pressure in the chamber. This will helps the gold adhere to the surface. Then, argon gas is pumped into the chamber to cleanse it. Finally, a thin layer of gold is spread across the surface, allowing the SEM to accurately read the surface of the polymer. After our samples were finally prepared, we could start viewing them under the microscope. This is where things got a bit boring. What I didn't know walking into this was how much time is spent waiting for the image to be created and how much more time is used looking for these tiny platelets. Hours. Many hours. This was slow and tedious and made me realize, once again, that patience is a virtue not granted to all (i.e., me). Looking for platelets only a few micrometers in diameter made the little discs seem endlessly large. It was like looking for a ring on a football field - possible, but time-consuming. We didn't even find any activated platelets. Although a bit frustrating (well, looking for hours for something that isn't there), this is good news! This means that the biodegradable polymer likely doesn't cause many platelets to activate (such as so many other surfaces do), reducing the risk of the polymer causing blood clots if used as a stent. While I don't have any cool pictures of activated platelets (because we couldn't find any), here is a cool something that we did find. Maybe a dust. Thoughts after this week:
Until next week! Maren
0 Comments
Leave a Reply. |
AuthorHello! I'm Maren, a high school senior and aspiring BME student. My goal is to share my experiences as I dive into the field of Biomedical Engineering. If you have any questions or remarks, please don't hesitate to leave a comment! Archives
April 2017
Categories |





 RSS Feed
RSS Feed
